Hoth Therapeutics (NASDAQ: $HOTH) is gaining fresh momentum in the microcap biotech space following its announcement of a key expansion in the CLEER-001 Phase 2a trial for HT-001. The company added Regis Clinical Research in Miami, Florida, as a new enrolling site, driven by patient demand outpacing current capacity, a clear sign of strong interest and operational progress.
Performance Snapshot
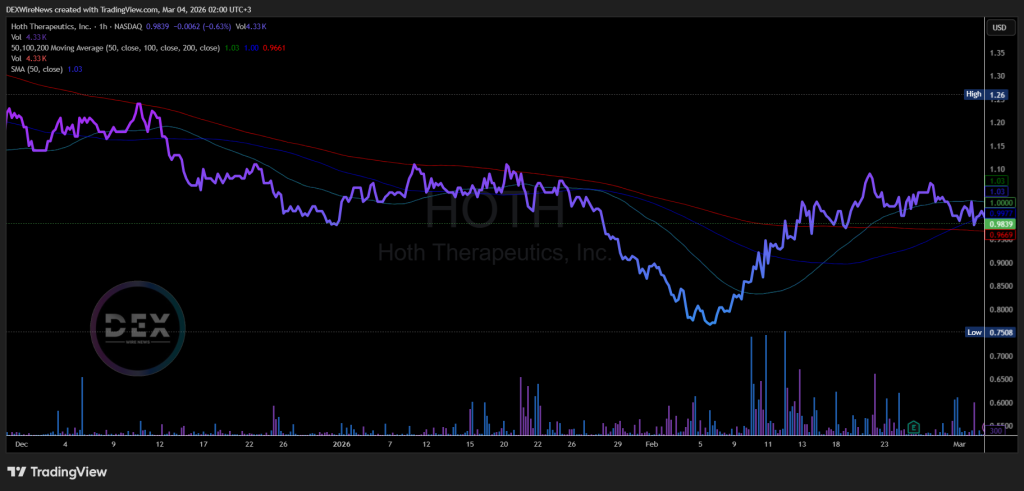
HOTH trades in classic microcap territory, where positive headlines and trial updates can spark rapid repricing. Recent pricing sits around $1.00 (with intraday fluctuations near $0.99–$1.02), within a 52-week range of $0.66–$2.12.
Year to date, HOTH is up 0.69%, while, the price is up 14.57% in the past 52 weeks, closing at $0.9968 on Tuesday, March 3, 2026. Over the past month, the stock has seen impressive gains of 18.81%.
Shares outstanding stand at approximately 15.51 million, yielding a market cap in the mid-teens (millions), and average daily volume hovers around 400K–450K shares, providing solid liquidity for quick moves on catalysts.
The Core Catalyst: HT-001 Enrollment Ramp
The new Miami site underscores Hoth’s push to accelerate enrollment in CLEER-001, targeting skin toxicities from EGFR inhibitor therapies in cancer patients. With no FDA-approved treatments specifically for this issue, and up to one-third of the roughly 200,000 annual U.S. patients facing dose reductions or discontinuations due to severe rash, itching, and lesions, HT-001 addresses a meaningful unmet need. Site additions like this are a practical accelerator, shortening the path to fuller datasets and potential milestones.
Encouraging Interim Signals
Hoth’s recent updates highlight promising early data driving physician and patient enthusiasm:
- 100% of open-label patients achieved the primary endpoint (ARIGA rash severity ≤1) by week six
- Zero EGFR inhibitor dose reductions or discontinuations tied to skin issues
- Over 65% reported significant pain/itching relief
- No dose-limiting toxicities observed
- A compelling case study (presented at AAD) showed complete resolution in one week for a metastatic breast cancer patient
These company-reported interim points, while early-stage, are the exact kind of signals that build visibility and momentum in small-cap biotech.
Added Tailwinds: IP and Pipeline Optionality
Hoth bolstered its portfolio with a U.S. Notice of Allowance for an exon-skipping patent in allergic diseases (targeting FC-Epsilon-RI-Beta and MS4A6A), enhancing IP strength and partnering potential. Separately, preclinical GDNF data in obesity/MASLD showed encouraging comparisons to semaglutide benchmarks, adding longer-term buzz in a hot therapeutic area, though still early.
What to Watch
Execution remains key: more site announcements, enrollment updates, detailed open-label data drops (targeted Q1 2026 per company timeline), IP issuances, and any financing moves to extend runway.
Bottom Line
HOTH is positioned as a catalyst-driven microcap with near-term focus on HT-001 progress. The new site addition signals real velocity in enrollment, backed by strong interim signals and a clean unmet-need story, setting the stage for potential upside as milestones approach.
Click Here for Updates on HOTH – It’s 100% FREE to Sign Up for Text Message Notifications!
DISCLAIMER — DEXWireNews has been compensated two thousand five hundred dollars electronically via bank wire by a third party for an advertising / marketing campaign for Hoth Therapeutics (NASDAQ: HOTH) beginning on 3/3/2026 and ending on 3/4/2026. This particular starter campaign will consist of just one email, one article, one push notification, one text message blast, a listing within our mobile app, and one informational mobile video distributed via social media. We may receive additional campaigns in the future directly from the company or by a third party to distribute media for Hoth Therapeutics (NASDAQ: HOTH). THIS IS NOT FINANCIAL ADVICE. Always do your own due diligence and consult a professional advisor. Trading stocks and cryptos involves significant risk of loss. Please read our Full Disclaimer: https://dexwirenews.com/disclaimer/